Death by Regulation
“How many of you have not been able to get a drug you needed to properly deliver anesthesia to a patient?” I asked.
Every hand in the room went up.
“How did that affect your patients?” I asked. “Two of our patients died,” one woman answered.
I was speaking to a group of nurse anesthetists, enrolled in a business management program at Marshall University in West Virginia. I wish I could say their experience is unusual. It isn’t.
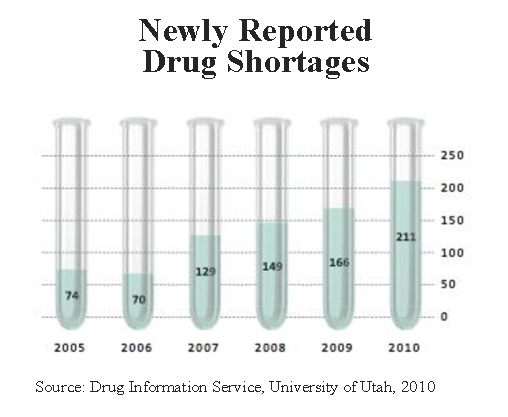
About 90 percent of all the anesthesiologists in the country report they are experiencing a shortage of at least one anesthetic. Drug shortages are also endangering cancer patients, heart attack victims, accident survivors and a host of other ill people. The vast majority involve injectable medications used mostly by medical centers, in emergency rooms, ICUs and cancer wards. Currently, there are about 246 drugs that are in short supply and, as the chart shows, the number has been growing for some time.
So what’s going on?
As early as 2005, hospitals and clinics complained to Health and Human Services Secretary Michael Leavitt that drug manufacturers and distributors were often out of certain drugs. The problem has been getting progressively worse ever since. A new report from the Premier healthcare alliance that found that drug shortages have risen to “critical levels,” endangering the public’s health. Hospitals are scrambling to make up the shortfall, in some cases rationing medications, postponing surgeries and using alternative drugs.
Industry insiders point to numerous causes of the problem, including the fact that the generic drug market may be inherently more volatile than the market for brand-name drugs. Imperfect competition may also be a factor. Others point to supply chain problems. Then there is government regulatory policy.
Output Controls. The Federal Food and Drug Administration (FDA) has been stepping up its quality enforcement efforts — levying fines and forcing manufacturers to retool their facilities both here and abroad. Not only has this more rigorous regulatory oversight slowed down production, the FDA’s “zero tolerance” regime is forcing manufacturers to abide by rules that are rigid, inflexible and unforgiving. For example, a drug manufacturer must get approval for how much of a drug it plans to produce, as well as the timeframe. If a shortage develops (because, say, the FDA shuts down a competitor’s plant), a drug manufacturer cannot increase its output of that drug without another round of approvals. Nor can it alter its timetable production (producing a shortage drug earlier than planned) without FDA approval.
Even the Drug Enforcement Agency (DEA) has a role — because minute quantities of controlled substances are often used to make other drugs. This is the apparent reason for a nationwide shortage of ADHD drugs, for example, including the generic version of Ritalin. And like the FDA, DEA regulations are rigid and inflexible. For example, if a shortage develops and the manufacturers have reached their preauthorized production cap, a manufacturer cannot respond by increasing output without going back to the DEA for approval.
The Centers for Medicare and Medicaid Services (CMS) has levied huge fines for “overcharging,” forcing some companies to leave the generic market altogether.
Price Controls. Also contributing to the problems of many facilities is a little known program that forces drug manufacturers to give discounts to certain end users. The federal 340B drug rebate program was created in 1992 to provide discounted drugs to hospitals and clinics that treat a high number of indigent patients, clinics treating patients on Medicaid, hospitals and clinics in the Public Health Service and certain Federally Qualified Health Centers (more listed here).Currently, the law requires drug companies to provide rebates of 23.1 percent for brand drugs; and 13 percent for generic drugs off of their average manufacturer’s price on qualifying outpatient drug use. States have the right to negotiate further discounts and actual rebates negotiated are typically much steeper than the federal requirement.
This state of affairs did not start with the Affordable Care Act (ObamaCare). By expanding the number of hospitals and clinics that are allowed to participate in the program, however, the Affordable Care Act will make things worse. In 2002, about 8,000 hospitals and clinics were in the program. By 2010 more than 14,457 were participating. The total number of eligible hospitals and clinics is now estimated at nearly 20,000.
Economics teaches that when prices are kept artificially low, shortages develop. People cannot get all of the care they try to obtain at the existing rate. Also, regardless of the apparently multiple causes of the shortages, certain patterns tend to emerge. People respond to persistent shortages by doing things that invariably make the problem worse.
Stockpiling. Buying organizations will typically respond by trying to stockpile quantities of drugs where supply is uncertain. That is, they will try to hoard more of the drugs than they ordinarily would keep in inventory in order to try to make sure they are available when needed. As the Healthcare Alliance Report explains, “drug shortages have been exacerbated by stockpiling on the part of providers,” who are trying to “protect themselves from the instability of the drug supply chain by placing orders that exceed normal requirements.”
Black Markets. Another thing that happens is the development of black (or gray) markets, where price gougers buy up quantities of a drug in short supply and sell it for a much higher price — even higher than would have been charged if the government had simply left the market alone. For example, in their 2005 letter to Secretary Leavitt, hospitals complained that shortages were exacerbated by drug distributors who filled their more lucrative commercial orders instead. (The federal government, incidentally, claims this is illegal.)
Members of the Premier healthcare alliance report paying “gray market” prices as much as 335 percent above the approved rate. A recent Kaiser Daily Health Policy Report highlighted how “the chain of custody in the gray market may pass from one distributor to another, creating a string of transactions that lead to higher prices and drug integrity concerns.”
Cascading Effects on Other Markets. Another consequence of shortages is that the effects in one market begin to cascade to other markets. In general, when hospitals cannot get a drug, they will turn to the next best alternative drug that creates the least adverse effects for patients. But as a Premier healthcare alliance analysis explains, when a shortage of one drug causes increased demand for a therapeutically similar product, the substitute product may also then be in short supply because it “is not normally produced in quantities sufficient to meet unanticipated market needs.” This scenario occurred last year with the morphine and subsequent hydromorphone shortages.
Solutions. Up till now, the Obama administration’s preference for regulation rather than market forces to solve safety problems is making the entire health care system less safe than it otherwise would have been.
Cass Sunstein, President Obama’s regulatory czar, announced last week that the administration intends to repeal cost-increasing, unnecessary regulations from 30 different agencies. If the administration is serious in this effort, a good place to start is with a web of regulations that are preventing life saving drugs from reaching the patients who need them.


I saw the version of this that was up at Health Affairs. Very good job.
The problem is bound to get worse. A decade ago, only about 8,000 facilities and clinics qualified for the discounted drug program. The Affordable Care Act extends the right to purchase drugs at steeply discounted priced to nearly 20,000. That means fewer customers paying market prices and more getting a bargain.
You should be ashamed for bringing economics into such a discussion…
Where is the song pairing. I feel cheated.
Very interesting post. And very worrisome.
We saw part of this phenomenon beautifully demonstrated when the flu vaccine came into short supply and a bad flu was predicted some years back. The way to manage such a shortage was to provide the flu vaccine to those that needed it most, hoping that the rest could be vaccinated later . There was fear in the air among the concentrated senior populations that were most at risk. They depended upon their physicians to supply them with a vaccine that was reimbursed by Medicare at very low levels. That meant that physicians most able to provide the vaccine to the most needy received only a small fraction of their vaccine request while the pharmacies, not burdened with the overhead, paperwork, etc. could pay a higher price for the vaccine and charge a higher price to those lined up for hours to get the shot. The pharmacies got the bulk of the initial vaccine supply.
With all the hype everyone lined up at the pharmacies. People lined up early in the day and waited for hours for their shot or until the pharmacy ran out of their supply. Who could stand on line for that long in inclement weather? Those that needed the shot least. The elderly that could not stand had to rely upon the inadequate supply many physicians were given. Areas, however without populations with strong need of the vaccine were given similar amounts of vaccines and in some of those areas as more vaccine flowed in they had more than necessary so some end users were shipping that vaccine to areas of need. A lot of people requiring the vaccine that year didn’t get it and many got it late. The sickest that could not fight their way through the lines frequently didn’t get vaccinated and sometimes at the pharmacy an ambulance would be summoned to pick up the senior that passed out in the heat.
This fiasco was created by government intervention into a place they do not belong. That was an early case of the problems we would be facing later, but government’s need to study problems for years or even decades before they can figure out solutions that are already out of date means that government is not the answer.
This is more than worrisome. It is scary.
No wonder I did not get the option of “going under” to get my upper wisdom teeth pulled. They highly encouraged local anesthesia. Wow did that hurt! I guess I did my part in this time of shortage.
Wow. What an eye opener. Conventional wisdom is that “Pharma” is the second most profitable industry in the world, oil being the first. And, that the FDA spends most of its time and resources approving new and improved drugs without proper trials so they can be churned out to the public at exorbitant prices. I have to say, I have no idea what’s true and what’s not. I have had many surgeries, the latest being two weeks ago, and I have never had any issues with drug shortages, and certainly not with anesthesia. In fact, as far as I can tell, I’ve always had cutting edge care, especially where surgery is concerned. I’m not wealthy, I don’t have a “Cadillac Health Plan”, and I don’t live in a particularly affluent demographic, as far as I can tell. I know that if anyone’s information can be trusted it’s Mr. Goodman’s, so I have to say I am truly flabbergasted.
This post is an eye opener.
I think someone should point out that the survey included Indonesia, Qatar, and Brazil, among other foreign countries. It lost its credibility with me after I read it. See below (from the original source):
The Drug Shortages Survey was administered online from April 12-27, 2011. The 1,373 anesthesiologist respondents represented 48 states (all but ND and WY), the District of Columbia, Puerto Rico and six nations (United States, Canada, Indonesia, Switzerland, Qatar and Brazil)
It’s a shame to see a shortage based solely on government policy (rather than supply chain disruptions or whatnot).
I’m a practicing anesthesiologist (physician). Patient above, claimed there were
“no drug shortages” at her facility. That may be an inaccurate statement since she would never know. These shortages are real and frequent. Over the past several years I have seen a number of useful medications we use during a typical general anesthetic exit the market, mostly d/t hospital’s bottom line. We adapt and just subsititute an older, less expensive, but safe medication. I don’t believe this negatively affects pt care. However, over last 12 months, we have experienced a very limited supply run of several medications that are necessary and need to be readily available. Both, older, cheap medications. One, a medication used to treat post-operative nausea, a very common side-effect and one that can increase costs d/t unecessary prolonged time in the recovery area and/or hospital admission. The second, a potentially life-saving muscle relaxant used frequently, especially in emergencies. Problem has been rectified, but if we are having these problems with older, less expensive meds, what about the newer ones. We all know about past shortages of flu vaccine, now the only manufacturer that makes anti-venom to coral snake bites, is on it’s last batch. I fear that additional regulation/price controls by an “all-knowing” federal bureaucracy like Obamacare will make things much worse.
It seems as though the FDA’s “zero tolerance” regime needs to be evaluated, it seems to be regulating indicators of quality rather than quality itself.
i am completely disgusted with the drug shortage that seems to happen every year hear in largo florida . i think that the state and locals love a pat on the back for closing the barn door after the fact. way to go florida. again . you have been ineffective in the past and using the same playbook again and again just wont work. so ??????????? you think that you would do better but the mensa club removed its residence somewhere else didn’t they. due to lack of ____, fill it in ! get a clue boys and girls.